Nano and Biomaterials, Manufacturing and Quality Control
The Nanomedicine Manufacturing Laboratory (NML) at Duquesne University, supported by multiple federal awards, develops and scales advanced nano- and biomaterial platforms for therapeutic delivery, multimodal imaging, and oxygen transport applications. The laboratory integrates materials science, pharmacology, and bioengineering to engineer reproducible, sterile formulations that meet rigorous quality and safety standards for both in vitro and in vivo studies.
The Nanomedicine Manufacturing Laboratory operates under implemented Quality-by-Design (QbD) principles, embedding controlled manufacturing workflows and predefined release specifications into every project. Each formulation undergoes comprehensive physicochemical and biological characterization—including stability, performance, and safety assessments—prior to distribution to collaborating institutions for translational evaluation. This integrated manufacturing-to-validation framework enables the development of targeted pain nanomedicines; oxygen-carrying nanoplatforms tailored for human tissue and organ machine-supported preservation; and multifunctional imaging agents for immune cell tracking across diverse disease and injury models.
Through sustained partnerships with federal agencies, industry collaborators, and national and international academic institutions, the Nanomedicine Manufacturing Laboratory advances innovation at the interface of nanotechnology, therapeutics, and regenerative medicine.
Research Areas
Nanomedicine Manufacturing Laboratory (NML) is a materials-focused research and manufacturing laboratory specializing in the sterile production of nanomaterials and biomaterials for imaging, drug delivery, and oxygen delivery. The Nanomedicine Manufacturing Laboratory manufactures nanoemulsions, microemulsions, micelles, colloidal dispersions, solid lipid nanoparticles, and hydrogel-based biomaterials, with routine production at the liter scale using controlled, reproducible processes.
All materials are produced under defined manufacturing workflows with embedded Quality-by-Design (QbD) principles. The Nanomedicine Manufacturing Laboratory conducts in-house material characterizations, pharmacological testing, and quality control, including batch-release testing prior to delivery to collaborating laboratories across the USA and abroad. Products are manufactured as sterile formulations when required and prepared for local, parenteral, or implantable use depending on the application.
The Nanomedicine Manufacturing Laboratory maintains a comprehensive product-tracking and quality-monitoring systems for all nanomedicine and biomaterial products developed under collaborative projects. Each product is uniquely coded, documented, tested, and released to collaborating laboratories only after a Certificate of Analysis (CoA) is issued. All collaborating sites are required to execute a Material Request Form, enabling continued quality control monitoring across the full product lifecycle—from manufacture through study completion and post-use assessment.
The Nanomedicine Manufacturing Laboratory products are rigorously evaluated for safety,
performance, and reproducibility across multiple preclinical models and collaborative
programs. This work is supported by the U.S. Air Force, Congressionally Directed Medical
Research Programs, National Institutes of Health, and ARPA-H, and involves partners
across academia, industry, and government.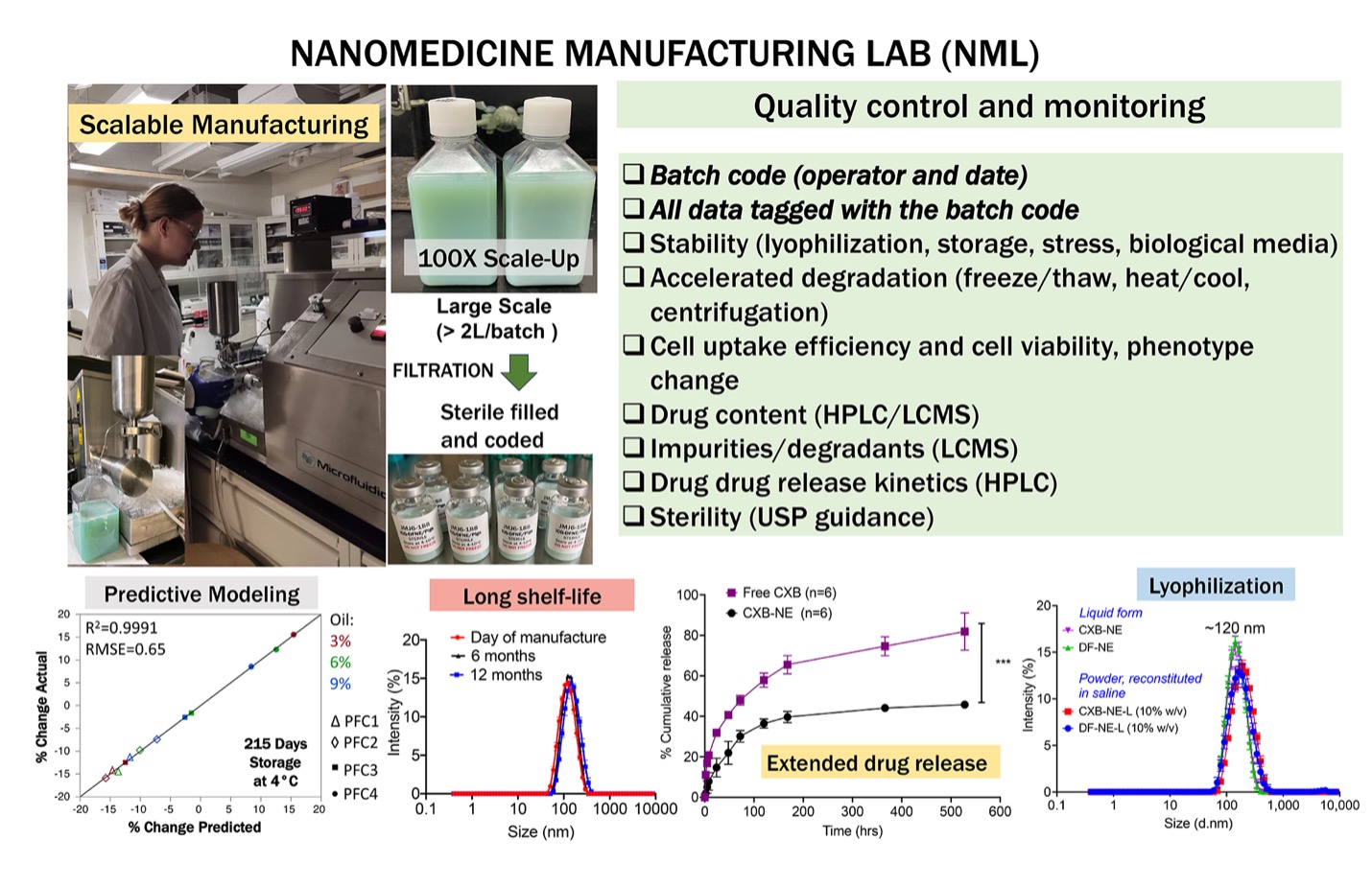
NML has also been a full MTEC Member since 2023.
Pain nanomedicine at the Nanomedicine Manufacturing Laboratory is an innovative, non-opioid
analgesic platform designed to support injury-specific targeted neuroimmunomodulation,
neuroregeneration, and neuromuscular recovery. The platform achieves targeted, sustained
modulation of neuroimmune interactions at the site of injury while minimizing systemic
exposure and avoiding side effects in the central nervous system, gastrointestinal
tract, and cardiovascular system. Non-invasive imaging is used to monitor the localization
and persistence of nanomedicine at the target site over time, enabling correlation
between biodistribution and functional outcomes, including neuromuscular recovery
and prolonged analgesia. To date, pain nanomedicine has been evaluated in numerous
preclinical models. Through partnerships with collaborating laboratories in neuroscience,
pain biology, and reconstructive surgery, pain nanomedicine has also contributed to
the discovery of new mechanisms in pain biology. Theranostic pain nanomedicine has
been successfully used as a nanoimaging probe to investigate pain mechanisms, particularly
the roles of immune cells. At Duquesne University, John A. Pollock, Professor of Neuroscience,
and his lab have used this platform to establish the role of immune cells in acute
and chronic neuropathic pain and to elucidate sex differences in pain biology.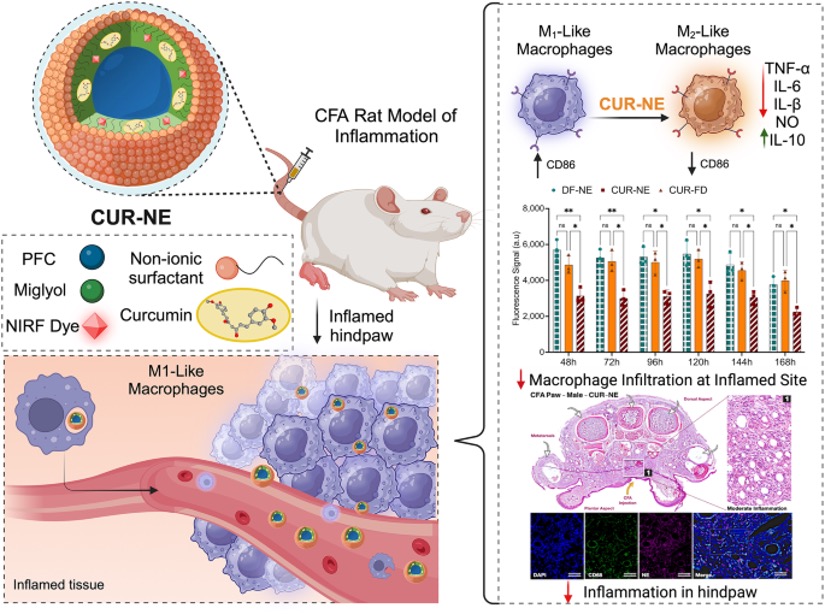
Image reproduced from: J Nanobiotechnol 23, 80 (2025). https://doi.org/10.1186/s12951-025-03164-w
Funding:
- W81XWH-20-1-0854: Long-Acting Non-Opioid Analgesic (LANA) Nanomedicine for Rapid, Single Dose, Sustained Pain Control and Rehabilitation Support After Neuromusculoskeletal Injury)
- AFMSA FA8650-17-2-6836: A Targeted, Local, Non-Opioid Therapeutic Strategy for Mitigation, Modulation, and Management of Trauma Induced Neuropathic Pain
- NIH NIDA R21 DA039621-01: Theranostic Pain Nanomedicines: imaging inflammation, reducing pain and need for opioids
- W81XWH-20-1-0276: Neuro-Immuno Modulating Analgesic (NIMA) Nanomedicine Platform for Treatment of Diabetic Neuropathy
Patents filed in 2025.
Oxygen-delivery materials developed at the Nanomedicine Manufacturing Laboratory are
based on perfluorocarbon nanoemulsions (PFC-NEs), which function as biocompatible,
acellular oxygen carriers with high oxygen solubility and chemical and metabolic inertness.
Unlike hemoglobin-based systems, oxygen delivery from perfluorocarbons is directly
governed by local oxygen partial pressure, enabling controlled delivery to hypoxic
tissues. The Nanomedicine Manufacturing Laboratory has developed stable, nanoscale
perfluorocarbon formulations that support oxygen delivery, alone or in combination
with therapeutic and imaging components, enabling extended (> 48h) metabolic support
and organ viability, and enabling real-time, non-invasive imaging of perfusate flow
during machine perfusion. For applications including machine-based tissue, limb, and
solid-organ preservation, these materials are engineered to access distal microvasculature,
maintain stability during extended perfusion, and enable non-invasive tracking of
perfusate distribution.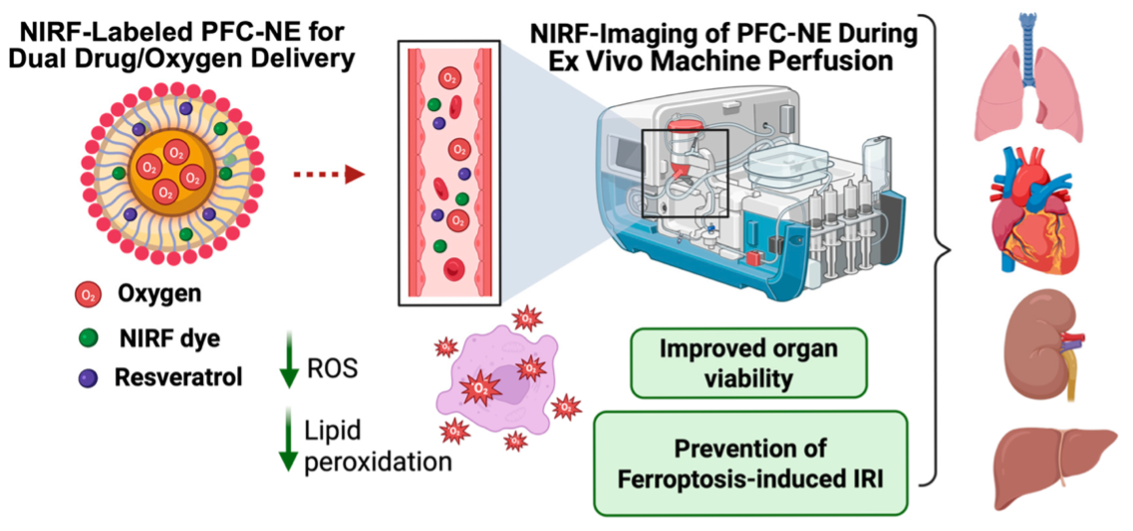
Image reproduced from Pharmaceutics 2026, 18, 143. https://doi.org/10.3390/pharmaceutics18020143
Funding:
- FA8650-20-2-6224: Semi-Autonomous Battlefield Resuscitation and Evacuation/Nano-Emulsion Oxygen Carrier for Advanced Resuscitation and Evacuation (NEOCARE)
- HT9425-24-1-0828: Resuscitation by Endothelial Stabilization and Targeted Oxygen Rescue (RESTOR) Platform for Vascularized Composite Allotransplantation
Patents filed in 2024.
Nanoimaging agents developed at the Nanomedicine Manufacturing Laboratory enable non-invasive,
longitudinal imaging of immune cells, with primary applications in neuroinflammation
and transplant rejection. These materials function as multimodal imaging platforms,
incorporating near-infrared fluorescence (NIRF) and magnetic resonance imaging (MRI)
capabilities to support spatial and temporal tracking of immune cell distribution
and dynamics in vivo. The Nanomedicine Manufacturing Laboratory nanoimaging agents
support longitudinal immunomonitoring in translational models across species, enabling
repeated, non-invasive assessment of immune cell infiltration, persistence, and spatial
organization over time. The materials are sterile, safe, non-immunogenic, and stable
in vivo for weeks to months, depending on biological context and study design. Nanoimaging
agents are routinely manufactured at large scale (>1 L per batch) using controlled,
reproducible processes to support multi-site collaborative studies.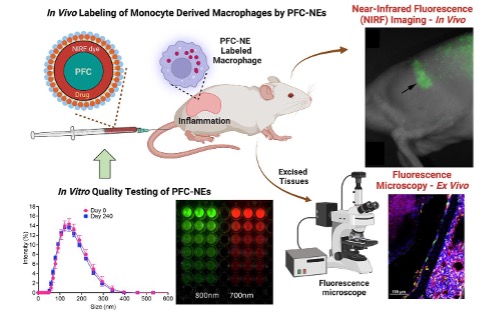
Image reproduced from: Bio-protocol 13(19): e4842. DOI: 10.21769/BioProtoc.4842.
Funding:
- W81XWH-20-1-730: Nanoimaging for Noninvasive Monitoring of Donor and Recipient Immune System Contribution to Acute and Chronic Rejection in VCA)
- W81XWH-19-1-0828: A Novel Graft Implanted “Macrophage-Enzyme Responsive Immunosuppressive Therapy” (MERIT) to Prevent Chronic Rejection in Vascularized Composite Allotransplantation)
- NIH NIBIB R21EB023104-01: Multimodal (PET/MR/NIR) imaging supported drug delivery in inflammatory diseases
The Nanomedicine Manufacturing Laboratory develops sterile nanosystem-loaded hydrogels
as biocompatible and non-immunogenic biomaterial platforms for localized therapeutic
delivery and immune modulation. These platforms include hydrogels loaded with nanosystems—for
example nanoemulsions (nanoemulgels), microemulsions (microemulgels), and micelles
(micelle-loaded hydrogels)—enabling localized, multi-payload presentation within a
single formulation. These hydrogel platforms are thermoresponsive and injectable,
allowing minimally invasive administration, and can be safely implanted into wounds
or surgical sites where in situ gelation or stabilization supports sustained local
activity. Sterile nanosystem-loaded hydrogels developed at the Nanomedicine Manufacturing
Laboratory are designed for wound healing, local multi-drug delivery, and targeted
modulation of immune cell activity at sites of injury or implantation.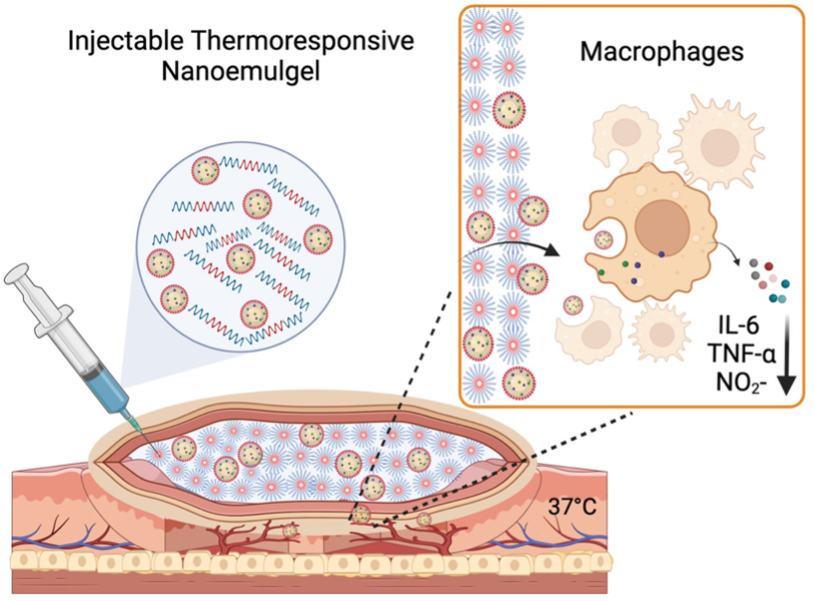
Image reproduced from Pharmaceutics 2023, 15, 2372. https://doi.org/10.3390/pharmaceutics15102372
Funding:
- AFMSA FA8650-17-2-6836: A Targeted, Local, Non-Opioid Therapeutic Strategy for Mitigation, Modulation, and Management of Trauma Induced Neuropathic Pain
- W81XWH-20-1-0276: Neuro-Immuno Modulating Analgesic (NIMA) Nanomedicine Platform for Treatment of Diabetic Neuropathy
- W81XWH-20-1-0769: Novel Graft Implanted Macrophage Targeted Nanoemulgels for In Situ Immunosuppression in Vascularized Composite Allografts
Collaboration
- Wake Forest School of Medicine
- BMI Organ Bank
- Stanford University
- Harvard Medical School
- Brigham and Women's Hospital
- The University of Utah
- Eversight
- Functional Circulation
- Moran Eye Center
- The Metis Foundation
- The University of Texas MD Anderson Cancer Center
- University of Pittsburgh
- Faron Pharmaceuticals
- UT Health San Antonio Long School of Medicine
- Joint Base San Antonio
- Naval Medical Research Unit San Antonio
- Carnegie Mellon University
- TDA Research
- inStem


